ABOUT US
Clearpoint Image Review Centre is an imaging CRO that provides sponsors with extensive medical, operational and image management expertise in support of clinical trials.
Our specialist team has successfully brought numerous clinical studies through US FDA and EU inspections with no major findings. Our method is based on proven SOPs adding value and speed to your clinical studies shortening your time to market.
Some key numbers:
Digitally imported images from 1000+ hospitals from all over the world.
Completed 100+ clinical studies.
Most of the studies we have supported were multimodality studies with 2-4 modalities.
Study set up fastest time recorded: 14 days from request to study start.
Study close fastest time recorded: 2 days from last subject to database closed.
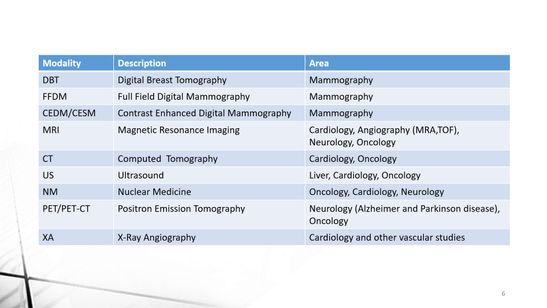
We support the following modalities:
DBT
FFDM
CESM
MRI
CT
Ultrasound
Nuclear Medicine
PET/CT
ECG
XA
Optical Imaging
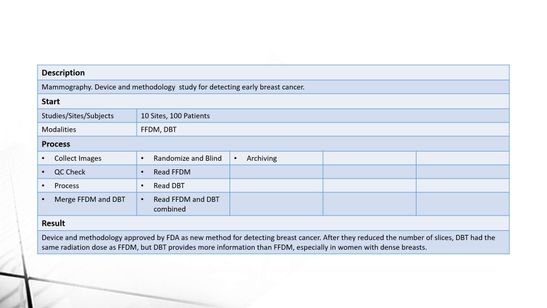
Some examples of what we have done